Melting point: the temperature at which a substance changes from a solid to a liquid at a given pressure water = 0oC Boiling point: the temperature at which a substance changes from a liquid to a gas at a given pressure water = 100oC Density is the amount of mass per unit of volume. Luster malleability: the ability to be hammered into a thin sheet ductility: the ability to be stretched into a wire Magnetism Electrical/thermal conductivity Solubility Solution = Solute + Solvent Solute: “stuff” being dissolved Solvent: “stuff” doing the dissolving The solvent is present in greater quantity The solute is present in the lesser quantity Ex: Salt water: Salt=solute, Water=solventġ1 How is it all related? Protons Electrons Neutrons Atoms Matterġ2 What are properties? Property: a characteristic of a substance that can be observed Physical property: a property that can be observed without changing the identity of the substance.

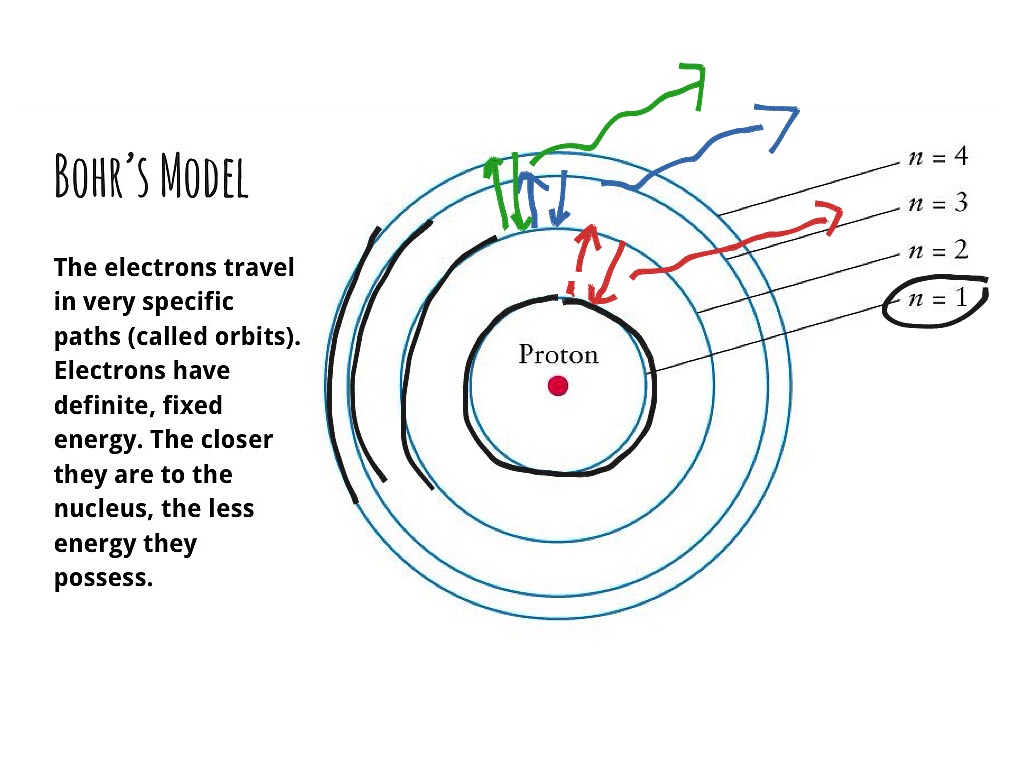
Covalent bond: formed when two atoms share electrons.Ĩ What is a compound? A compound is a pure substance that is created by 2 or more elements chemically reacting and joining together Ex: NaCl, H2O, CO2, NH3, NaHCO3, and C6H12O6 Compounds form to allow elements to become more stable Na is flammable when it comes in contact with H2O, and Cl2 is a toxic gas NaCl is a very stable compound that is neither flammable nor toxic (in normal quantities)ĩ What is a mixture? A mixture is the physical combination of 2 or more substances It is important to understand that a mixture is not chemically combined Mixtures can be separated by physical means such as filtration, distillation, and chromatography Remember no chemical change is occurringġ0 What is a mixture? Homogeneous mixtures are commonly called solutions. Ion: an atom that has gained or lost one of more electrons. Ionic bonds: bonds formed by the transfer of electrons. Valence Electrons: electrons in the outermost energy level of an atom. Nucleus: central core made of protons and neutrons. Proton: a particle with a positive charge.ħ Compounds Neutron: neutral particle, does not have a charge.Įlectron: has a negative charge. Atom: the smallest unit of an element that has all the properties of the element. Chemical Formula: uses chemical symbols and subscripts to identify the number of atoms of each element in a molecule of a compound. Compound: a type of matter that forms when two or more elements combine chemically.Ħ Compounds Molecule: the smallest unit of a compound that has all the properties of the compound. Mixture: a type of matter that forms when two or more substances are combined but do not join together chemically. Synthetic element: elements that are made by scientists in a laboratory and do not exist in nature. Element: a substance that cannot be broken down into simpler substances by ordinary chemical means. Matter: anything that has mass and volume. Synthetic Chemical: a chemical that is not formed in nature and is made by people. Example: Carbon C Hydrogen H Oxygen O Carbon C Iron Fe Sodium Na Magnesium MgĤ Identify Chemicals Chemical: substance used in or formed by a chemical process. Example: Carbon Cģ List common elements & chemical symbols.Ĭhemistry List common elements & chemical symbols. Example: Carbon CĬhemistry List common elements & chemical symbols. Presentation on theme: "Chemistry Draw a Bohr Model of a carbon atom."- Presentation transcript:ġ Chemistry Draw a Bohr Model of a carbon atom.Ģ List common elements & chemical symbols.

